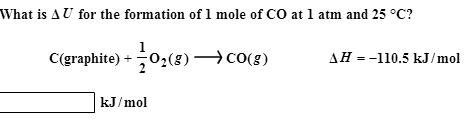Calculate the number of moles in(i) 8 g of oxygen atoms(ii) 4.088 × 10^21 number of oxygen atoms (Atomic mass of O = 16 u )
A mixture of 2 mole of CO and 1 mol of O2 is ignited. Correct relationship is (1)△ H=△ U (2)△ H >△ U (3)△ H

Chapter 8 “Stoichiometry” Mr. Mole. Section 8.2 The Arithmetic of Equations u OBJECTIVES: Interpret balanced chemical equations in terms of: a) moles, - ppt download

UMM.com@6周年ありがとう on Twitter: "U-Mol Festival 〜全夜祭〜 日程:2018/08/10 (金) 出演者:U- Mol(ゆとり、もるでお) ゲスト出演者:ATY,いりぽん先生,りりり,カケリネ&伊礼亮 チケットURL:https://t.co/TTgTzCg7yf https://t.co/Z924K75zjH" / Twitter

SOLVED:Convert each to mass in grams. a. 4.22 ×10^15 atoms U b. 8.65 ×10^25 atoms H c. 1.25 ×10^22 atoms O d. 4.44 ×10^23 atoms Pb
![Calculate value of -U/100, for AB(s), from following data of Born-Haber's cycle. [where U is lattice energy in kJ/mol] Calculate value of -U/100, for AB(s), from following data of Born-Haber's cycle. [where U is lattice energy in kJ/mol]](https://d10lpgp6xz60nq.cloudfront.net/physics_images/BLJ_VKJ_ORG_CHE_C01_E07_005_Q01.png)
Calculate value of -U/100, for AB(s), from following data of Born-Haber's cycle. [where U is lattice energy in kJ/mol]