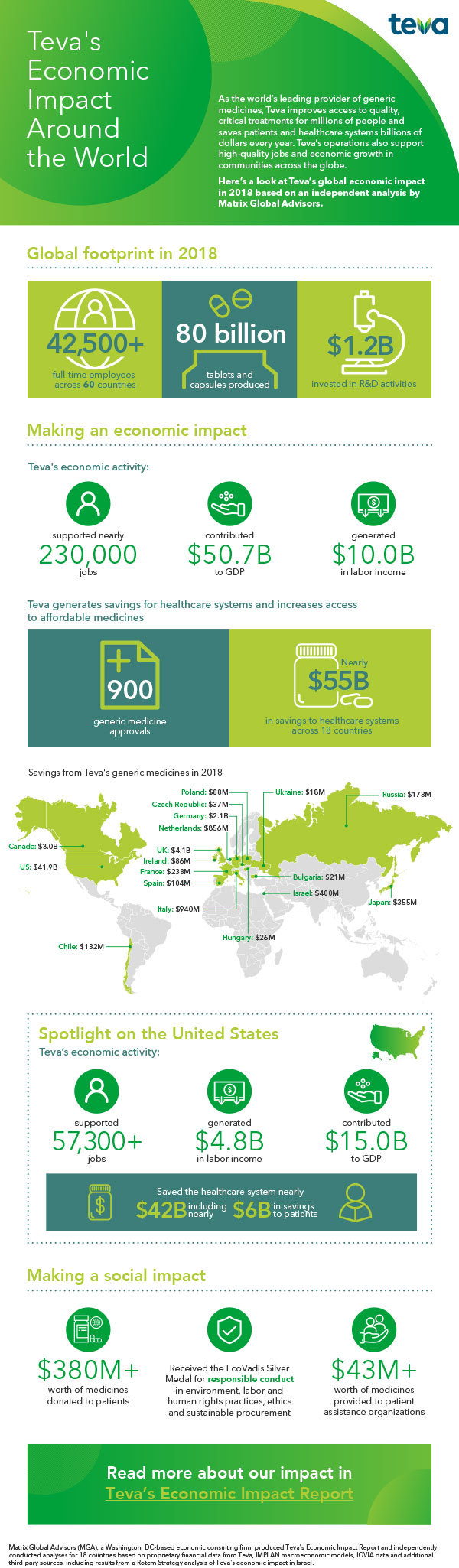
Teva Pharmaceuticals USA Issues Voluntary Nationwide Recall of all Amlodipine/Valsartan Combination Tablets and Amlodipine/Valsartan/Hydrochlorothiazide Combination Tablets that are Within Expiry | FDA

Teva Announces Largest-Ever Trial to Collect Information from Patients and Physicians About TD, its Impact and Progression Over Time, and Evaluate Treatment-Related Outcomes | Business Wire

Teva Pharmaceuticals Announces Launch of Daily Maintenance Asthma Medication | Asthma and Allergy Foundation of America