Stereoselective synthesis of (1R, 2S)-norephedrine by recombinant whole-cell biocatalysts coupling acetohydroxyacid synthase I and ω-transaminase - ScienceDirect

Investigation of the l-phenylacetylcarbinol process to substituted benzaldehydes of interest - ScienceDirect
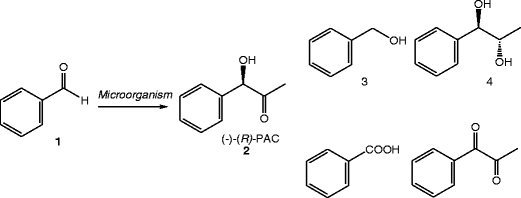
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink
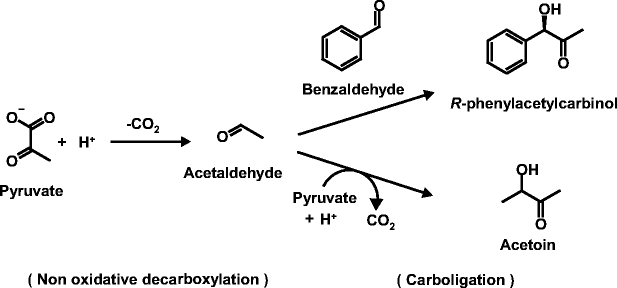
Enzymatic (R)-phenylacetylcarbinol production in a benzaldehyde emulsion system with Candida utilis cells | SpringerLink

Improvement of the yeast based (R)-phenylacetylcarbinol production process via reduction of by-product formation - ScienceDirect
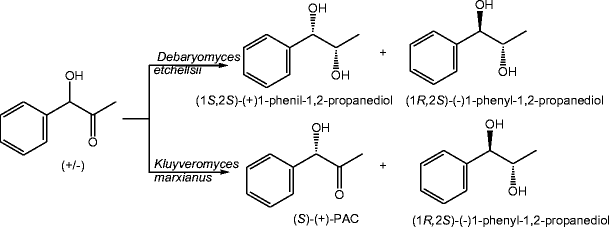
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink

An N-methyltransferase from Ephedra sinica catalyzing the formation of ephedrine and pseudoephedrine enables microbial phenylalkylamine production - ScienceDirect

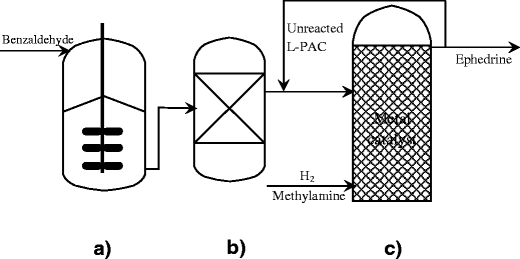
Biotransformation of benzaldehyde to L‐phenylacetylcarbinol (L‐PAC) by Torulaspora delbrueckii and conversion to ephedrine by microwave radiation | Semantic Scholar