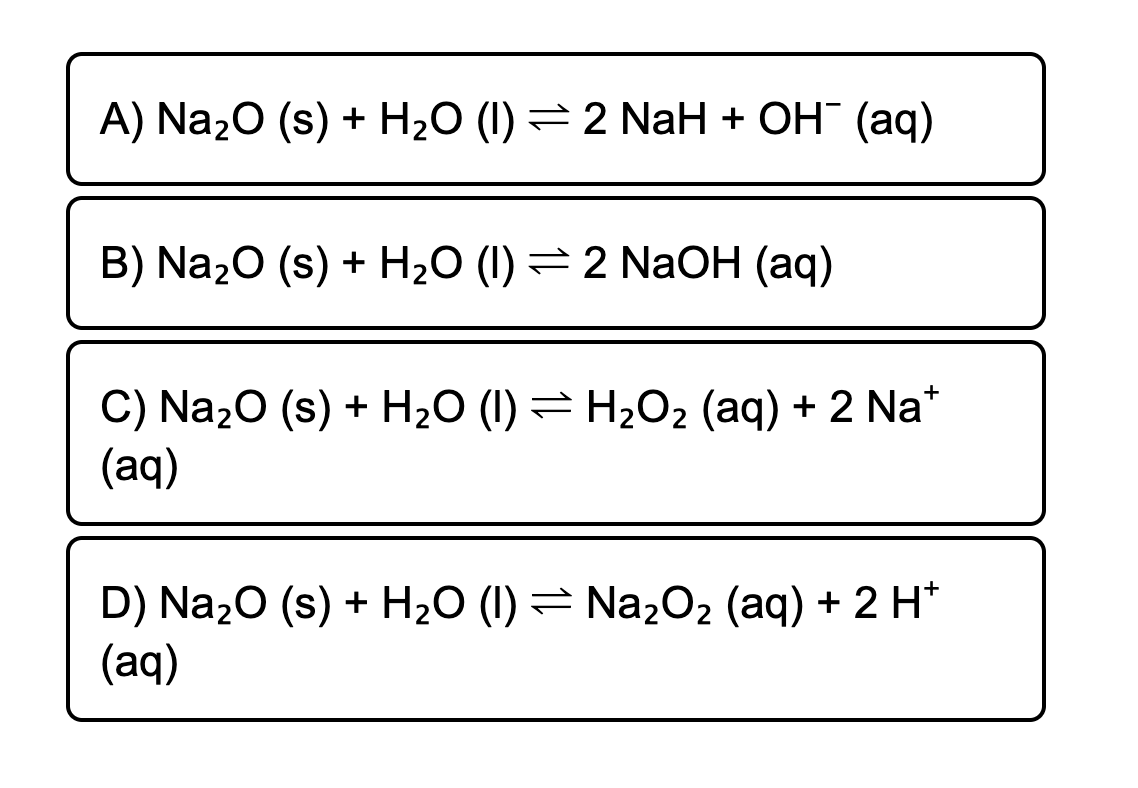
SOLVED: Na2O+H2O =NaOH cantidad de atonomos antes de balancear Na2O+H2O =NaOH cantidad de atonomos después de balancear

Chemical Reactions Balancing Chemical Reactions. Chemical Reactions Objectives List three observations that suggest that a chemical reaction has taken. - ppt download

Materials | Free Full-Text | Alkali Activation of Waste Clay Bricks: Influence of The Silica Modulus, SiO2/Na2O, H2O/Na2O Molar Ratio, and Liquid/Solid Ratio

SOLVED: Part A Translate the formulaic equations t0 word equa tions. Na2o H2O NaOH 2HgO(s) 2Hg(l) 02(g) BaCi2(aq) Na2CrO4aq) BaCrO4(s) 2NaCllaq) CS2() 302(g) cO2(g) 2502(g) NaCllaq) AgNO3(aq) NaNO3aq) AgCIls) Part B Translate
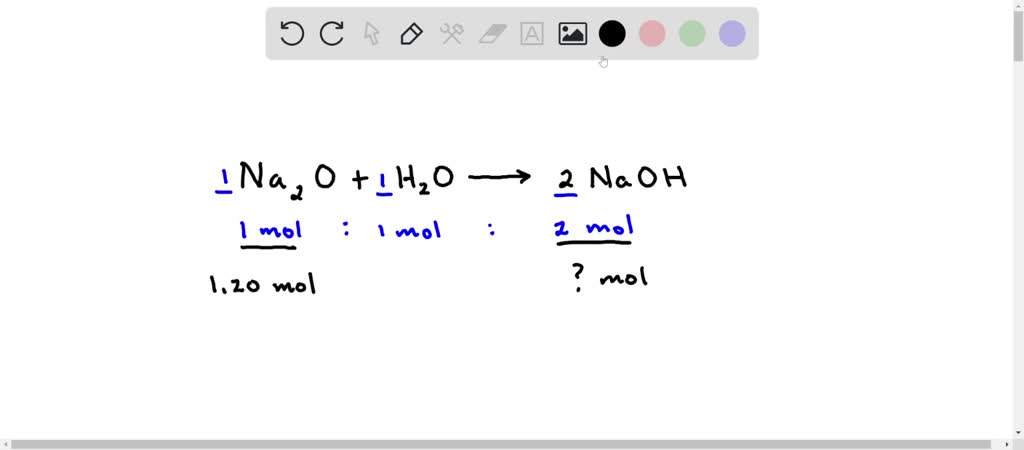
SOLVED: Given the following equation: Na2O + H2O —> 2 NaOH How many moles of NaOH are produced from 1.20 moles of Na2O?
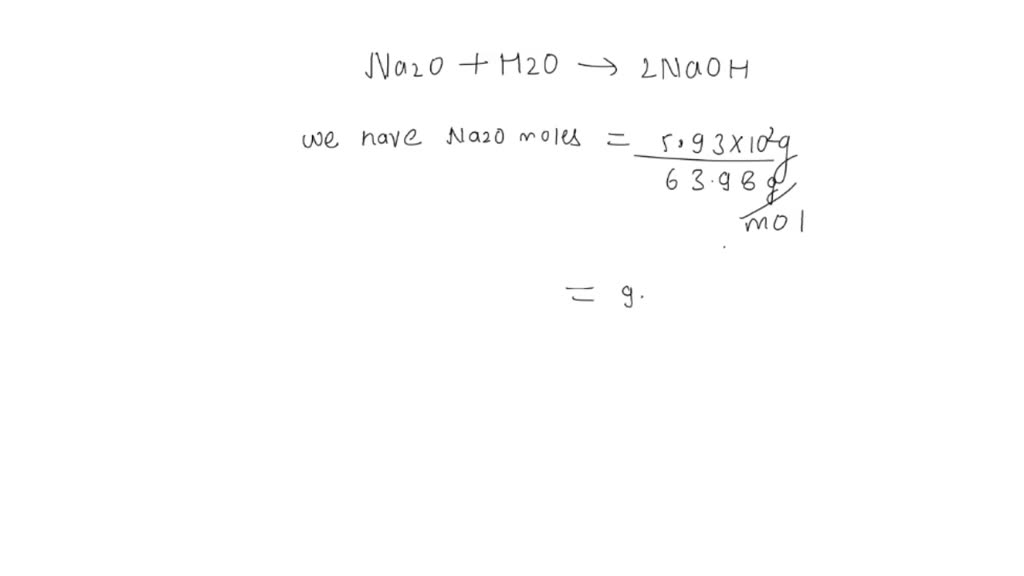
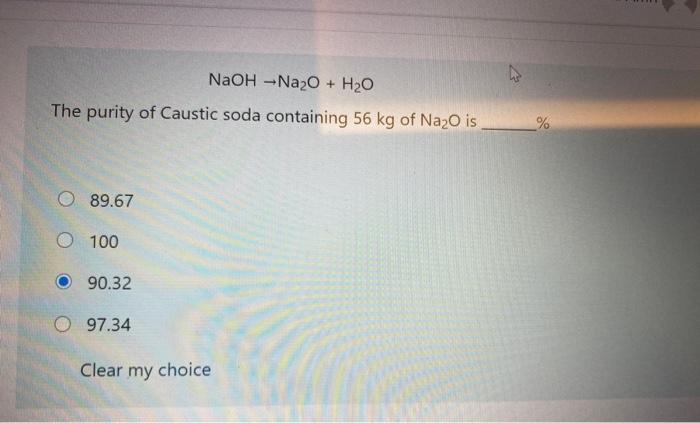
SOLVED: 3. Given the following unbalanced reaction: Na2O + H2O —> NaOH How many grams of NaOH is produced from 5.93 x 102 grams of Na2O?

3. Balance the following equations a CH4 +O2 = CO₂ + H₂Ob Na2O +H2O=NaOHc CA(OH)2+HCI=CaCI2+H2Od - Brainly.in

How to balance Na2O+H2O=NaOH|Chemical equation Na2O+H2O=NaOH|Na2O+H2O=NaOH balance equation - YouTube