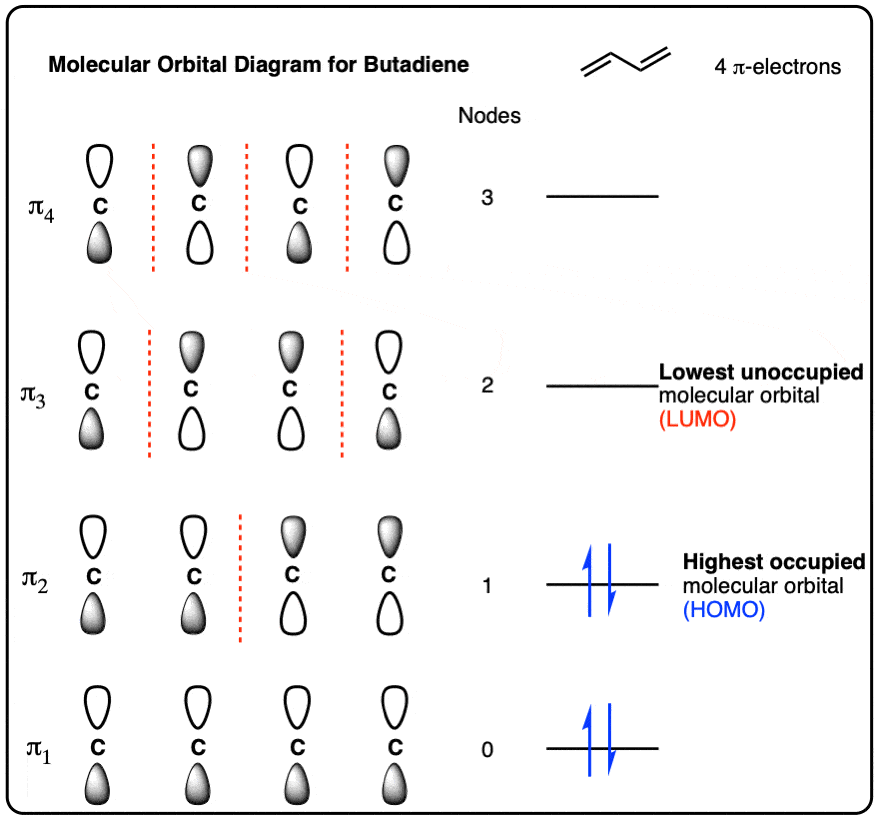
Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are

Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are
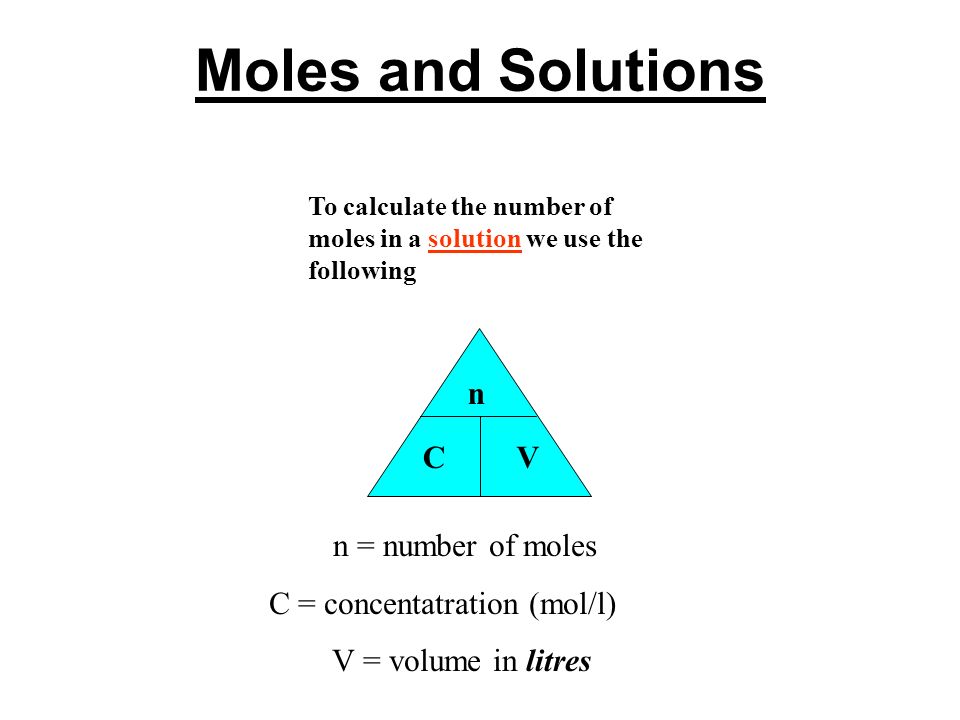
Moles and Solutions g n gfm To calculate the number of moles in a solution we use the following n CV n = number of moles C = concentatration (mol/l) V. - ppt download

141 Mole Worksheet Key - Mass and Moles Worksheet How many moles in 34 g of sulfur? 34 푔 푆 × 1 푚표푙 푆 - Studocu

SOLVED: A compound of carbon and hydrogen contains 92.3% C and has a molar mass of 78.1 g/mol. Select all that is correct. A. The empirical formula is CH. B. The molecular









:max_bytes(150000):strip_icc()/vitamin-c-molecular-model-483948223-582c8a523df78c6f6a473f1c.jpg)