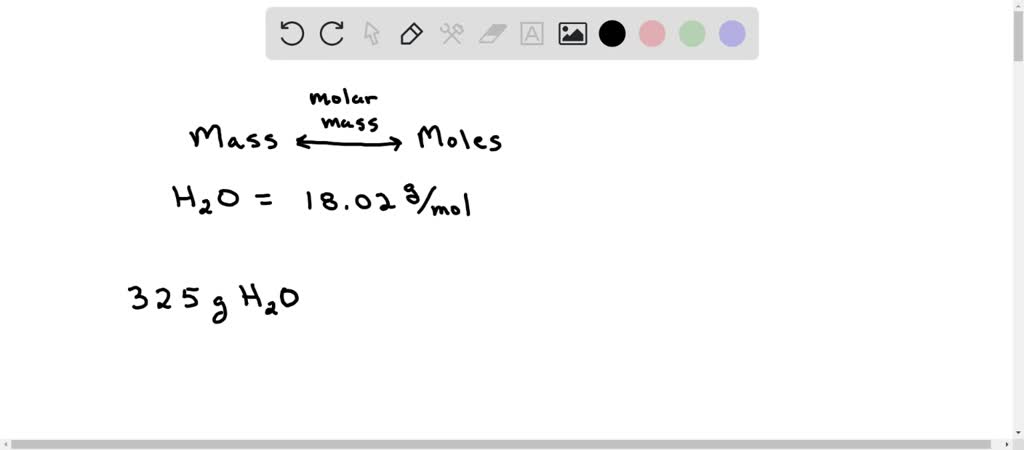
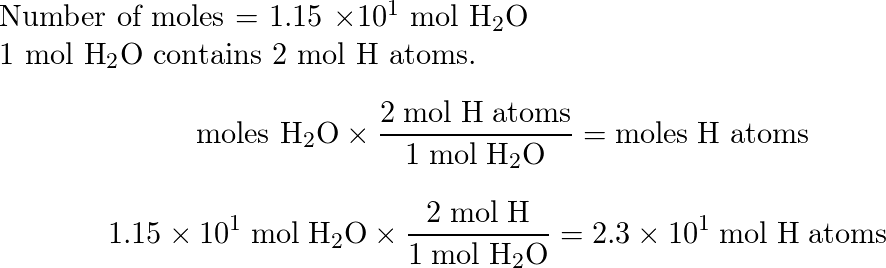Question Video: Determining the Mass of Water Lost When a Hydrated Compound of Cobalt(II) Sulfate Is Heated | Nagwa

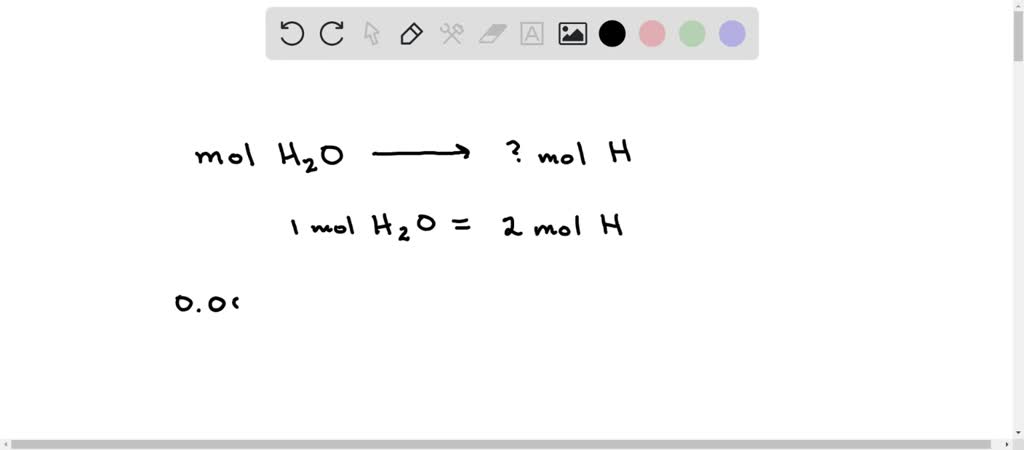
SOLVED: How many moles of hydrogen atoms are present in a sample that contains 3.64 moles of water, H2O? answer=mol H How many moles of H2O molecules are present in a sample

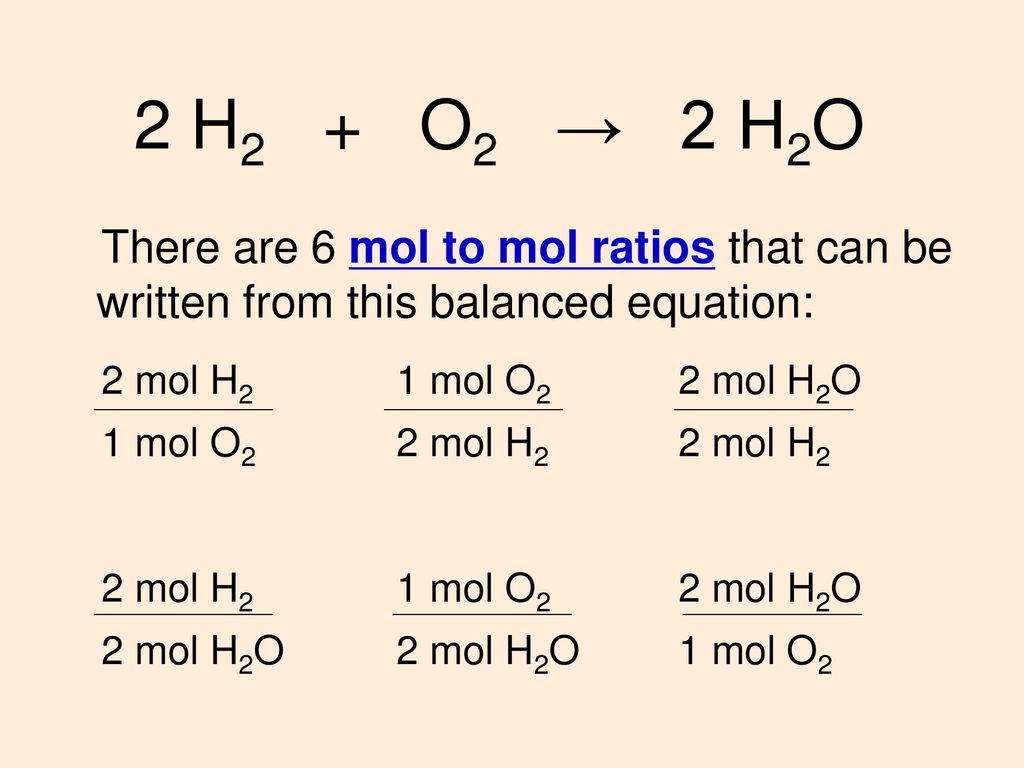
Solved: 9 mol P4010 reacts with 51 mol H₂O according to the equation below: P4010 + 6H₂O → 4H3PO4 How many - Brainly.com