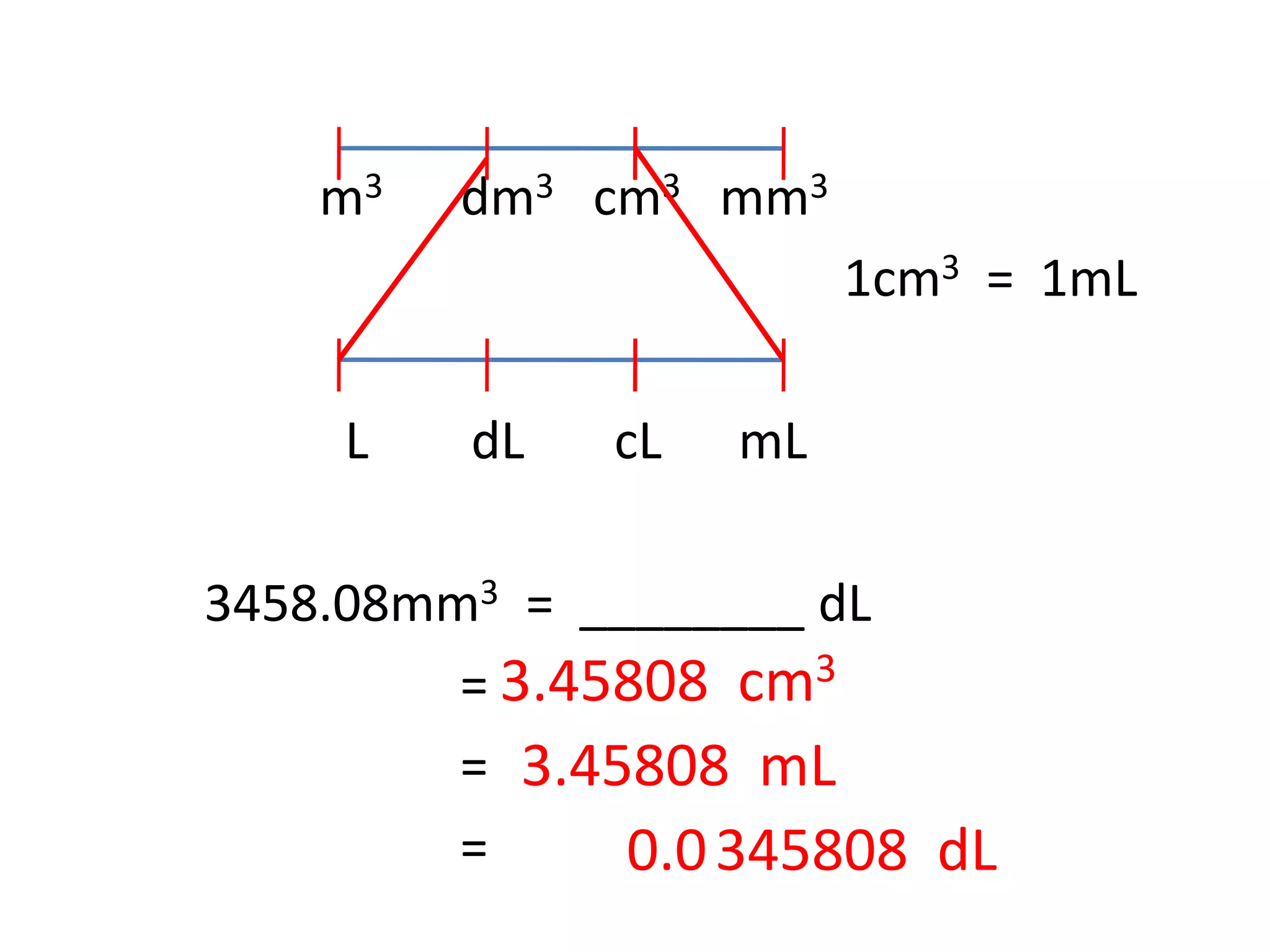
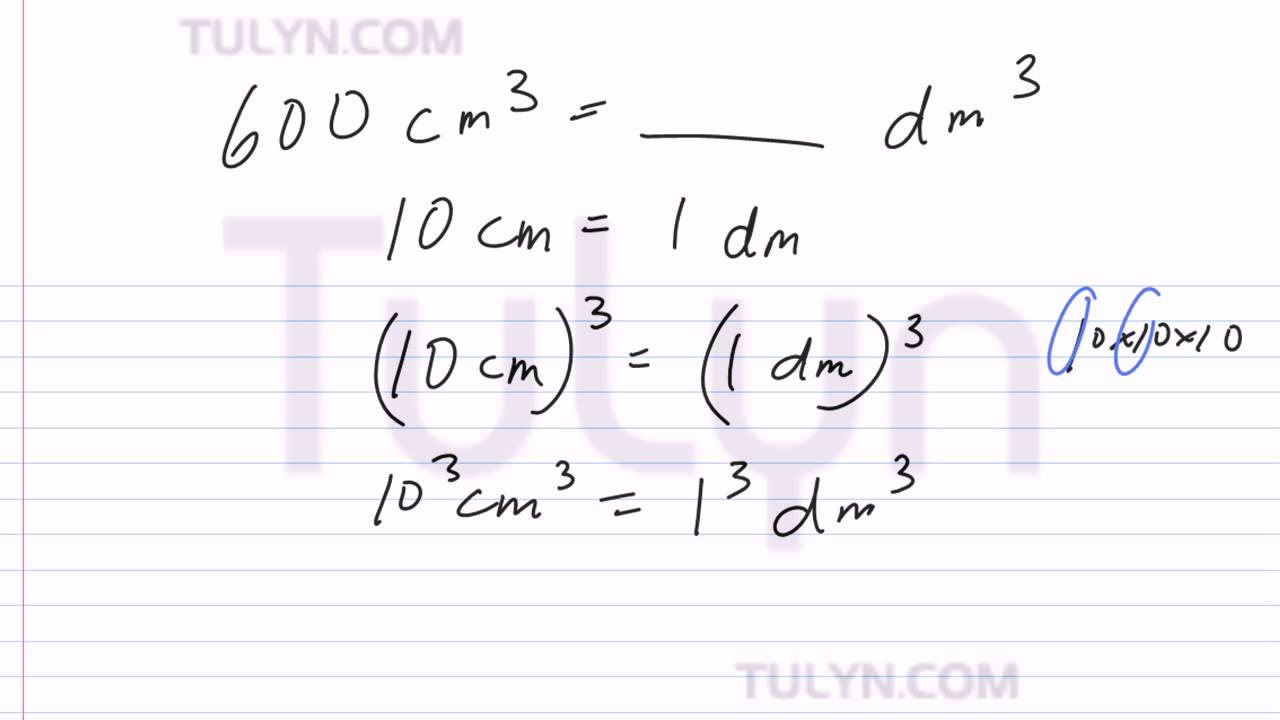SOLVED: Q1 If molarity of Na2CO3is 0.05 M at a volume of 25.00 cm3 while the titre value of HClis 14.00 cm3, calculate the concentration of HCIin molldm3. Equation of the reaction

Energies | Free Full-Text | A New Control Method for a Bi-Directional Phase-Shift-Controlled DC-DC Converter with an Extended Load Range

Amount of Substance in Relation to Volumes of Gases (3.5.1) | AQA GCSE Chemistry Revision Notes 2018 | Save My Exams

Visible-Light-Driven Photocatalytic H2 Production from H2O Boosted by Hydroxyl Groups on Alumina | Industrial & Engineering Chemistry Research

Concentrated aqueous sulphuric acid is 98% H2SO4 by mass and has a density of 1.80 gm L^-1 . What is the volume of acid required to make one litre of 0.1 M H2SO4 solution?

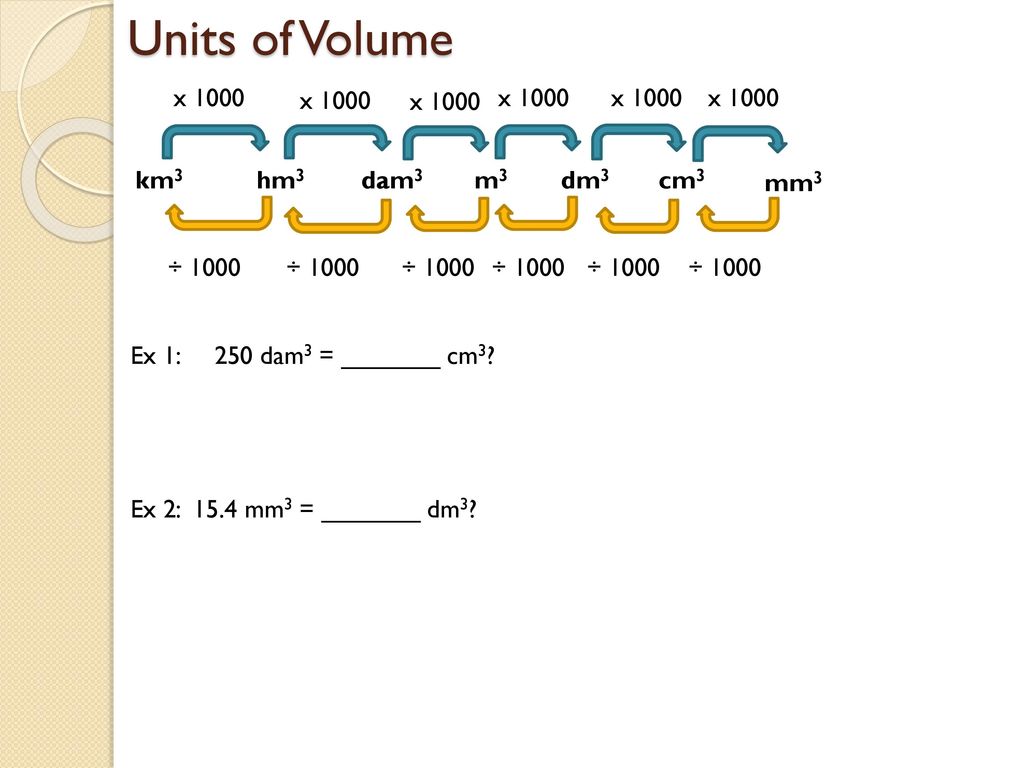
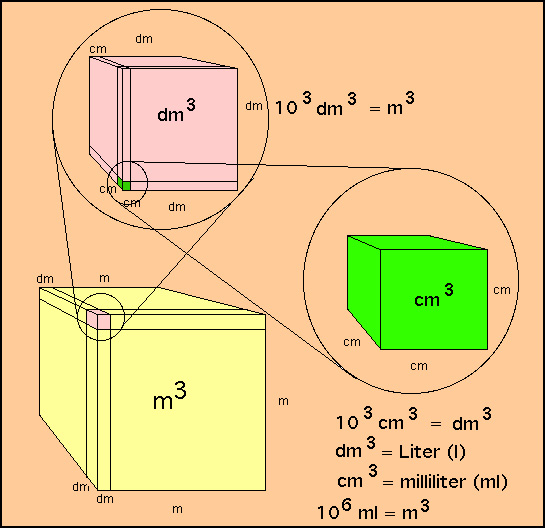
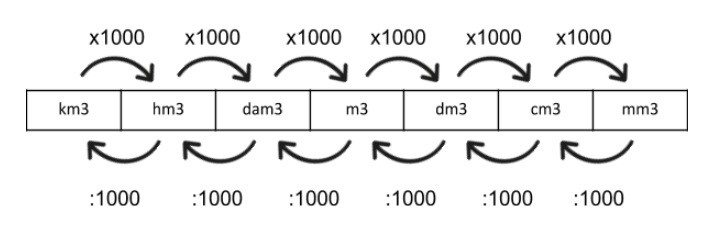
Try This!OOsoFill in the blanks.a 1 litre =b 5 litres =millilitresdm3-cm3- dm3• cm3litresc 10,000 cm3 - Brainly.in